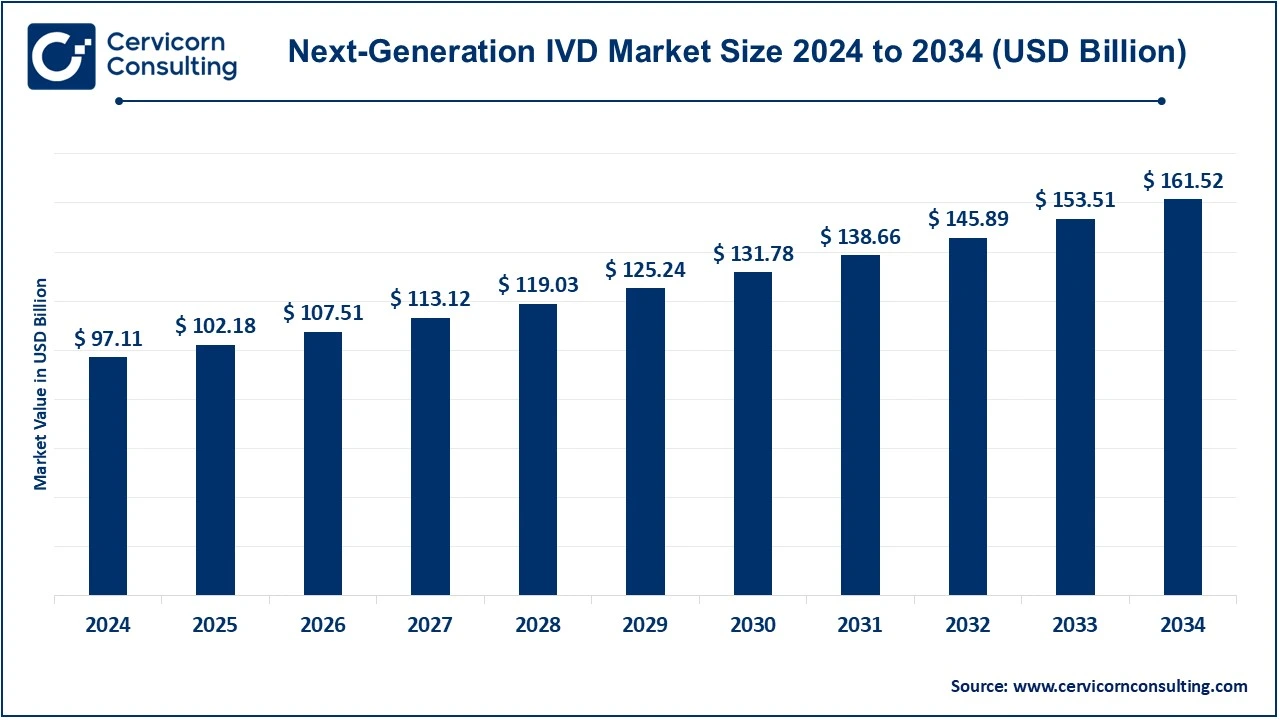
The global next-generation IVD market size was valued at USD 97.11 billion in 2024 and is expected to be worth around USD 161.52 billion by 2034, growing at a compound annual growth rate (CAGR) of 5.21% during the forecast period 2025 to 2034.
Growing fast, the next-generation IVD market comes from an increased need for modern diagnostic tools, more so in some areas such as cancer diagnosis, infectious disease testing, and personalized medicine. Further growth will be a result of various factors contributing to the world's aging population, increasing instances of chronic illnesses, improvement in molecular diagnostics technology, and emphasis on early disease detection and prevention. The constant transition into point-of-care testing and the incorporation of AI in diagnostics is driving the market further.
In vitro diagnostics (IVD) is defined as the examination of samples obtained directly from the human body outside the human body in a controlled environment such as a laboratory. These devices use in vitro diagnostics for the detection, diagnosis, and monitoring of infections and diseases of patients. All these tests encompass a range of technologies like reagents, instruments, as well as software. Used as a system, this provides crucial information about the health status of a patient. IVD is of extreme importance for clinical decision-making by helping the healthcare personnel understand the presence, absence, or progression of a medical condition.
CEO Statements
Robert B. Ford, CEO of Abbott
"At Abbott, we are committed to advancing healthcare by pioneering innovative, next-generation diagnostic solutions that empower clinicians and patients alike. Our new-generation IVD technologies will provide faster, more accurate, and more accessible testing, helping to transform the way diseases are detected and managed. We’re excited about the possibilities these breakthroughs hold to improve patient outcomes and drive meaningful progress in global healthcare."
Alexandre Merieux, CEO of bioMérieux SA
“At bioMérieux, we are convinced that, only by¯taking into account our entire ecosystem and the¯public interest, will we be able to succeed in building a healthier world and a more inclusive society”.
Bernd Montag, CEO of Siemens Healthineers AG
"At Siemens Healthineers, we are committed to pioneering the next generation of in-vitro diagnostics, leveraging cutting-edge technology and AI to empower clinicians with more accurate, faster, and accessible diagnostic tools. Our goal is to enhance patient outcomes globally and drive a transformation in healthcare that prioritizes precision and efficiency."
Report Scope
Report Scope
| Area of Focus | Details |
| Market Size in 2024 | USD 97.11 Billion |
| Projected Market Size in 2034 | USD 161.52 Billion |
| CAGR 2025 to 2034 | 5.21% |
| Dominant Region | North America |
| Accelerating Region | Asia-Pacific |
| Key Segments | Type, Product, Application, End User, Region |
| Key Companies | Abbott, bioMérieux SA, QuidelOrtho Corporation, Siemens Healthineers AG, Bio-Rad Laboratories, Inc., Qiagen, Sysmex Corporation, Charles River Laboratories, Quest Diagnostics Incorporated, Agilent Technologies, Inc., Danaher Corporation, F. Hoffmann-La Roche Ltd. |
The next-generation IVD market is segmented into type, product, application, end user, and region. Based on type, the market is classified into core laboratory diagnostics, point of care testing, and molecular diagnostics. Based on product, the market is classified into consumables, instruments, and software. Based on application, the market is classified into oncology, infectious diseases, diabetes, cardiology, and other. Based on end user, the market is classified into academic & research institutions, diagnostic laboratories, hospitals & clinics, and others.
Core Laboratory Diagnostics: The core laboratory diagnostics segment has dominated the market in 2024. Advances in technology allow for the execution of core laboratory diagnostics in centralized facilities equipped with modalities such as PCR, NGS, and automated systems. They do everything from routine blood work to complex molecular analyses. In many instances, samples have to be processed centrally, and detailed diagnostic results obtained for diseases such as cancer, genetic disorders, or infections. These, therefore, form the core of the application of next-gen IVD technologies that necessitate specific equipment and expertise.
Point of Care Testing: In simple terms, POC testing refers to a diagnosis at or near the patient's location; for instance, a test done in a home or a clinic. The cornerstone of POC testing remains pregnancy tests and glucose meters. POC testing has gained popularity because it offers quick, cost-effective, and easy diagnostics that the consumer is demanding. Other innovations also include portable sensors and handheld PCR instrumentation that make possible the swift diagnosis of heart conditions, diabetes, infectious infections, and many others. Improved patient care is a byproduct of decisions being made at the speed with which they are now being made.
Molecular Diagnostics: Molecular diagnostics encompasses the techniques of diagnosing at the molecular level through any genetic material, such as DNA, RNA, or other forms of genetic material. One can detect genes carrying mutations, pathogens, and biomarkers with ultimate accuracy using techniques such as PCR and next-generation sequencing. This technology has become essential in personalized medicine, oncology, and infectious diseases. Early detection and disease monitoring will be important to build tailored treatment plans. That's why molecular diagnostics is being pushed toward the center stage of next-gen IVD technologies.
Oncology/Cancer: The oncology/cancer segment has dominated the market in 2024. Next-generation IVD technologies belong to the oncology area, where early diagnosis of cancer is related to genetic profiling and supervision of anticancer treatment. The identified mutations are associated with cancerous growth, and their rate can be monitored non-invasively. Molecular diagnostics in cancer will become, step by step, an imperative for treatments via targeted therapies and immunotherapies, putting oncology at the top position when talking about next-gen IVD solutions.
Infectious Diseases: Rapid identification of pathogens that cause infections revolutionizes molecular diagnostics. These pathogens include viruses and fungi, and indeed various types of bacteria. Infected patients can be rapidly diagnosed using polymerase chain reaction and sequencing technologies. The control of prompt therapies further advances because of this. Rapid diagnoses have improved during the COVID-19 pandemic. It is still evolving with enhancements towards the diagnosis of novel infectious threats and antibiotic-resistant bacteria.
Diabetes: This relies on continuous glucose monitoring and molecular diagnostics in the care for diabetes improvement. They measure real-time glucose levels and enable patients to manage the disease condition better. Risk factors for complications of diabetes are indicated through genetic testing. Innovation is driving next-gen IVD tools for the monitoring of diabetes, which delivers more personal and proactive care.
Cardiology: New-generation IVD technologies in cardiology aid in cardiovascular disease diagnosis through the identification of new sensitivity biomarkers such as troponin, along with other genetic tests. The diagnostics can detect the conditions much earlier than ever-including myocardial infarction, heart failure, and arrhythmias. Genetic profiles and cardiac biomarkers result in better therapy that is more customized and leads to better results for patients.
Others: Other than the core area of oncology, infectious diseases, diabetes, and cardiology, the horizon IVDs are to be applied in neurology, maternal health, and genetics testing. Molecular diagnostics have been applied in neurodegenerative diseases such as Alzheimer's as well as in non-invasive prenatal testing-giving preliminary clues into fatal health issues. This versatility is opening the range of applications for IVD technology to a horizon that can include various medical fields.
The Next-generation IVD market is segmented into several key regions: North America, Europe, Asia-Pacific, and LAMEA (Latin America, Middle East, and Africa). Here’s an in-depth look at each region
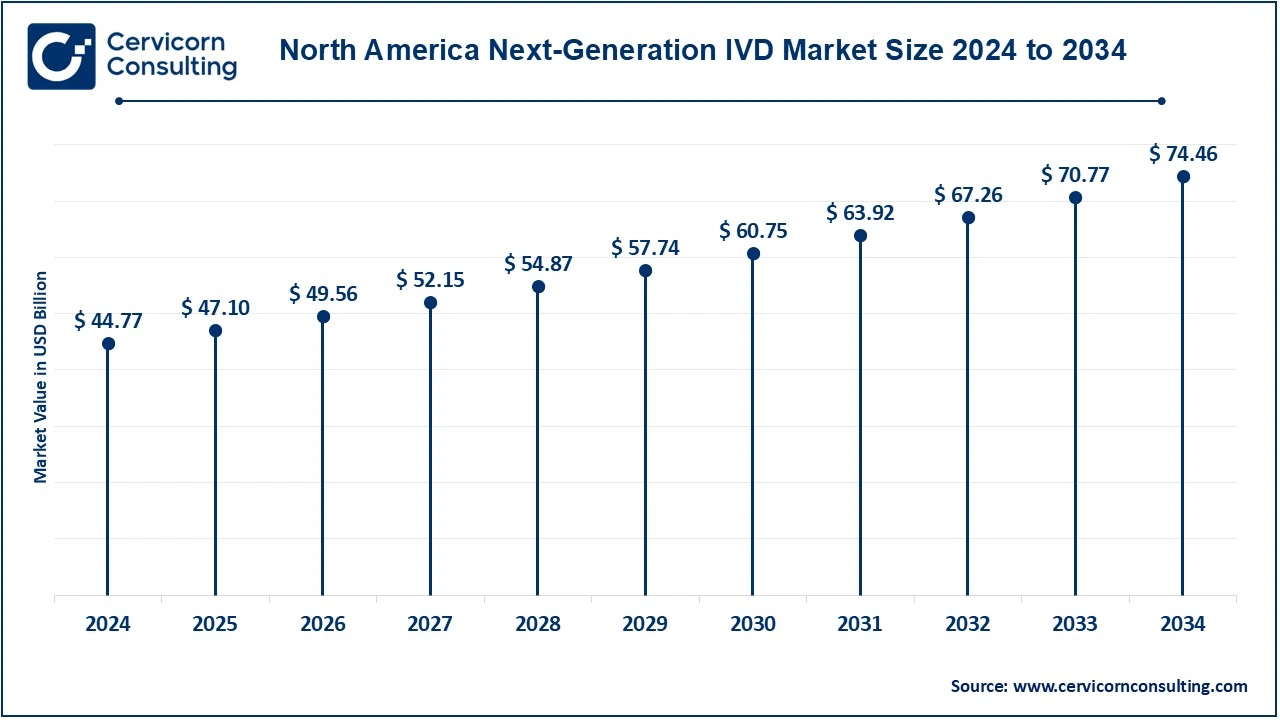
The North America next-generation IVD market size was valued at USD 97.11 billion in 2024 and is expected to be worth around USD 161.52 billion by 2034. North American countries within that region targeted are the United States and Canada because of mature healthcare infrastructure, technological innovation, and high expenditure on healthcare. The U.S. houses major biotechnology companies and a dynamic ecosystem for health care for next-generation diagnostic technologies, molecular diagnostics, next-generation sequencing, and point-of-care testing. Canada also had an excellent healthcare system focused on precision medicine, which had been rapidly increasing demand for advanced diagnostic solutions, especially in oncology, cardiology, and infectious diseases. Favorable reimbursement policies and stricter regulatory frameworks further accelerate market growth in North America. Another country with strong healthcare systems, the focus here is precision medicine. In this context, the region demands innovative advanced diagnostic solutions, mainly in oncology, cardiology, and infectious diseases. Favorable reimbursement policies combined with a strong regulatory framework work in favor of the growth of the North American market.
The Europe next-generation IVD market size was valued at USD 97.11 billion in 2024 and is expected to be worth around USD 161.52 billion by 2034. The Europe is also a large marketplace. Among European countries, Germany, the UK, France, and Italy stand on top of the list. These countries boast excellent healthcare systems, medical technology investment, and rising demand for personalized medicine. For instance, Avacta Group plc, October 2022, the clinical-stage oncology drug company whose product platforms are known by Affimer and preCISION, recently signed the acquisition of Launch Diagnostics Holdings Limited for a consideration of USD 25 million. Moreover, the acquirement would hold an earn-out of up to USD 13 million dependent on performance. According to the group, the acquirement represents the start of Avacta's M&A-driven growth strategy for its diagnostics division to establish an internationally integrated business for immunodiagnostics and molecular diagnostics. The company will exploit a fractured EU and UK market by buying firms that have complementary technology platforms to strengthen R&D and develop Avacta's diagnostics portfolio. Both the UK and France have public health systems, but much more developed, allowing for a broad uptake of sophisticated IVD solutions.
The Asia-Pacific next-generation IVD market size was valued at USD 97.11 billion in 2024 and is expected to be worth around USD 161.52 billion by 2034. Rapid growth is being seen in the still-emerging market in the APAC region due to increased healthcare needs, improvement in infrastructure, and growth in disposable incomes. India and China, with massive population bases and an increasingly growing healthcare system, are the two biggest markets to support the rise in demand for diagnostics, particularly in cases of chronic diseases such as diabetes, cancer, and cardiovascular disorders. Example: More than 100 million people in India are diabetic A new study published in the Lancet puts the number of people living with diabetes in India at 101 million - nearly 11.4% of the country's population. A survey commissioned by the health ministry also found that 136 million people - or 15.3% of the people - could be living with pre-diabetes. Type 2 is the most prevalent of this disease, wherein patients suffer from high blood sugar either due to their inability to produce sufficient amounts of insulin, the hormone, or not responding well to it. The leaders in terms of tech in healthcare with higher spending on healthcare and great innovation in diagnostic tools like NGS and molecular diagnostics, are Japan and South Korea.
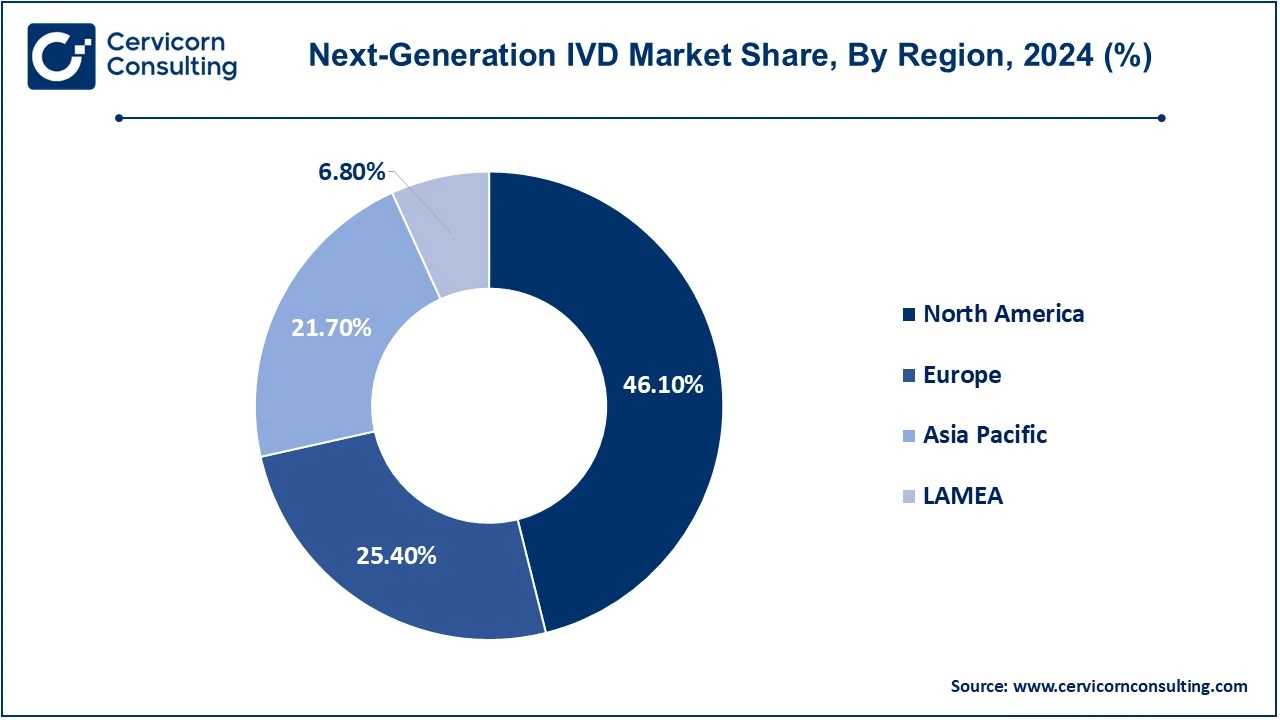
The LAMEA next-generation IVD market size was valued at USD 97.11 billion in 2024 and is expected to be worth around USD 161.52 billion by 2034. The adoption of IVD technology is slowly gaining pace and, currently, the LAMEA region Latin America, Middle East, and Africa maximum importance. Investment in health infrastructure of countries like Brazil, Mexico, and Argentina across communities of Latin American regions propel demands for advanced diagnostic tools, primarily by chronic patients with diabetes and cancer. Initiatives are underway in every nook and corner of the Middle East region, too--Saudi Arabia, United Arab Emirates, and Qatar among them, as the need for accurate diagnostics and targeted medicine grows. Thus, with an ever-growing point of interest towards integration of access and infrastructure around healthcare on the continent, diagnostics in Africa can prove to be extremely inexpensive, at least for infectious diseases.
Some of the recent new strategies and partnerships in the next-generation in vitro diagnostics (IVD) sector are driving innovation and partnerships significantly between leading industry players such as Abbott, bioMérieux SA, QuidelOrtho Corporation, Siemens Healthineers AG, and Bio-Rad Laboratories, Inc. These companies are leading the edge of diagnostic technology with real-time testing solutions through AI-driven platforms, molecular diagnostics, and point-of-care devices. Through partnering and acquisition, the combination of technologies for automated diagnostics, genomics, and microfluidics further energize to ensure speed, accuracy, and accessibility for diagnostics both from clinical and non-clinical sites. It is sure that in this dynamic IVD sector, the growing rate of innovation and collaboration underscores the need for even quicker, more precise, and customized solutions.
Some notable examples of key developments in the market include:
Market Segmentation
By Type
By Product
By Application
By End User
By Region
Chapter 1. Market Introduction and Overview
1.1 Market Definition and Scope
1.1.1 Overview of Next-Generation IVD
1.1.2 Scope of the Study
1.1.3 Research Timeframe
1.2 Research Methodology and Approach
1.2.1 Methodology Overview
1.2.2 Data Sources and Validation
1.2.3 Key Assumptions and Limitations
Chapter 2. Executive Summary
2.1 Market Highlights and Snapshot
2.2 Key Insights by Segments
2.2.1 By Product Overview
2.2.2 By Type Overview
2.2.3 By Application Overview
2.2.4 By End User Overview
2.3 Competitive Overview
Chapter 3. Global Impact Analysis
3.1 COVID 19 Impact on Next-Generation IVD Market
3.1.1 COVID-19 Landscape: Pre and Post COVID Analysis
3.1.2 COVID 19 Impact: Global Major Government Policy
3.1.3 Market Trends and Opportunities in the COVID-19 Landscape
3.2 Russia-Ukraine Conflict: Global Market Implications
3.3 Regulatory and Policy Changes Impacting Global Markets
Chapter 4. Market Dynamics and Trends
4.1 Market Dynamics
4.1.1 Market Drivers
4.1.1.1 Technological Innovation
4.1.1.2 Rising Healthcare Costs
4.1.1.3 Public health needs
4.1.2 Market Restraints
4.1.2.1 High Development Costs
4.1.2.2 Limited Reimbursement
4.1.2.3 Integration Challenges with Other Systems
4.1.3 Market Challenges
4.1.3.1 Delayed Approval by Regulatory Agencies
4.1.3.2 Supply Chain Disruptions
4.1.3.3 Intellectual Property Concerns
4.1.4 Market Opportunities
4.1.4.1 Increased test sensitivity and accuracy
4.1.4.2 Personalized medicine
4.1.4.3 Global Health Applications
4.2 Market Trends
Chapter 5. Premium Insights and Analysis
5.1 Global Next-Generation IVD Market Dynamics, Impact Analysis
5.2 Porter’s Five Forces Analysis
5.2.1 Bargaining Power of Suppliers
5.2.2 Bargaining Power of Buyers
5.2.3 Threat of Substitute Products
5.2.4 Rivalry among Existing Firms
5.2.5 Threat of New Entrants
5.3 PESTEL Analysis
5.4 Value Chain Analysis
5.5 Product Pricing Analysis
5.6 Vendor Landscape
5.6.1 List of Buyers
5.6.2 List of Suppliers
Chapter 6. Next-Generation IVD Market, By Product
6.1 Global Next-Generation IVD Market Snapshot, By Product
6.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
6.1.1.1 Consumables
6.1.1.2 Instruments
6.1.1.3 Software
Chapter 7. Next-Generation IVD Market, By Type
7.1 Global Next-Generation IVD Market Snapshot, By Type
7.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
7.1.1.1 Core Laboratory Diagnostics
7.1.1.2 Point of Care Testing
7.1.1.3 Molecular Diagnostics
Chapter 8. Next-Generation IVD Market, By Application
8.1 Global Next-Generation IVD Market Snapshot, By Application
8.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
8.1.1.1 Oncology/Cancer
8.1.1.2 Infectious Diseases
8.1.1.3 Diabetes
8.1.1.4 Cardiology
8.1.1.5 Others
Chapter 9. Next-Generation IVD Market, By Vehicle Type
9.1 Global Next-Generation IVD Market Snapshot, By Vehicle Type
9.1.1 Market Revenue (($Billion) and Growth Rate (%), 2022-2034
9.1.1.1 Academic & Research Institutions
9.1.1.2 Diagnostic Laboratories
9.1.1.3 Hospitals & Clinics
9.1.1.4 Others
Chapter 10. Next-Generation IVD Market, By Region
10.1 Overview
10.2 Next-Generation IVD Market Revenue Share, By Region 2024 (%)
10.3 Global Next-Generation IVD Market, By Region
10.3.1 Market Size and Forecast
10.4 North America
10.4.1 North America Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.4.2 Market Size and Forecast
10.4.3 North America Next-Generation IVD Market, By Country
10.4.4 U.S.
10.4.4.1 U.S. Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.4.4.2 Market Size and Forecast
10.4.4.3 U.S. Market Segmental Analysis
10.4.5 Canada
10.4.5.1 Canada Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.4.5.2 Market Size and Forecast
10.4.5.3 Canada Market Segmental Analysis
10.4.6 Mexico
10.4.6.1 Mexico Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.4.6.2 Market Size and Forecast
10.4.6.3 Mexico Market Segmental Analysis
10.5 Europe
10.5.1 Europe Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.5.2 Market Size and Forecast
10.5.3 Europe Next-Generation IVD Market, By Country
10.5.4 UK
10.5.4.1 UK Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.5.4.2 Market Size and Forecast
10.5.4.3 UKMarket Segmental Analysis
10.5.5 France
10.5.5.1 France Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.5.5.2 Market Size and Forecast
10.5.5.3 FranceMarket Segmental Analysis
10.5.6 Germany
10.5.6.1 Germany Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.5.6.2 Market Size and Forecast
10.5.6.3 GermanyMarket Segmental Analysis
10.5.7 Rest of Europe
10.5.7.1 Rest of Europe Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.5.7.2 Market Size and Forecast
10.5.7.3 Rest of EuropeMarket Segmental Analysis
10.6 Asia Pacific
10.6.1 Asia Pacific Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.6.2 Market Size and Forecast
10.6.3 Asia Pacific Next-Generation IVD Market, By Country
10.6.4 China
10.6.4.1 China Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.6.4.2 Market Size and Forecast
10.6.4.3 ChinaMarket Segmental Analysis
10.6.5 Japan
10.6.5.1 Japan Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.6.5.2 Market Size and Forecast
10.6.5.3 JapanMarket Segmental Analysis
10.6.6 India
10.6.6.1 India Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.6.6.2 Market Size and Forecast
10.6.6.3 IndiaMarket Segmental Analysis
10.6.7 Australia
10.6.7.1 Australia Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.6.7.2 Market Size and Forecast
10.6.7.3 AustraliaMarket Segmental Analysis
10.6.8 Rest of Asia Pacific
10.6.8.1 Rest of Asia Pacific Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.6.8.2 Market Size and Forecast
10.6.8.3 Rest of Asia PacificMarket Segmental Analysis
10.7 LAMEA
10.7.1 LAMEA Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.7.2 Market Size and Forecast
10.7.3 LAMEA Next-Generation IVD Market, By Country
10.7.4 GCC
10.7.4.1 GCC Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.7.4.2 Market Size and Forecast
10.7.4.3 GCCMarket Segmental Analysis
10.7.5 Africa
10.7.5.1 Africa Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.7.5.2 Market Size and Forecast
10.7.5.3 AfricaMarket Segmental Analysis
10.7.6 Brazil
10.7.6.1 Brazil Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.7.6.2 Market Size and Forecast
10.7.6.3 BrazilMarket Segmental Analysis
10.7.7 Rest of LAMEA
10.7.7.1 Rest of LAMEA Next-Generation IVD Market Revenue, 2022-2034 ($Billion)
10.7.7.2 Market Size and Forecast
10.7.7.3 Rest of LAMEAMarket Segmental Analysis
Chapter 11. Competitive Landscape
11.1 Competitor Strategic Analysis
11.1.1 Top Player Positioning/Market Share Analysis
11.1.2 Top Winning Strategies, By Company, 2022-2024
11.1.3 Competitive Analysis By Revenue, 2022-2024
11.2 Recent Developments by the Market Contributors (2024)
Chapter 12. Company Profiles
12.1 Abbott
12.1.1 Company Snapshot
12.1.2 Company and Business Overview
12.1.3 Financial KPIs
12.1.4 Product/Service Portfolio
12.1.5 Strategic Growth
12.1.6 Global Footprints
12.1.7 Recent Development
12.1.8 SWOT Analysis
12.2 bioMérieux SA
12.3 QuidelOrtho Corporation
12.4 Siemens Healthineers AG
12.5 Bio-Rad Laboratories, Inc.
12.6 Qiagen
12.7 Sysmex Corporation
12.8 Quest Diagnostics Incorporated
12.9 Charles River Laboratories
12.10 Agilent Technologies, Inc.
12.11 Danaher Corporation
12.12 F. Hoffmann-La Roche Ltd.